Patient in Georgia, Health Worker in New York Suffer Anaphylaxis After Getting Pfizer/BioNTech COVID-19 Vaccine
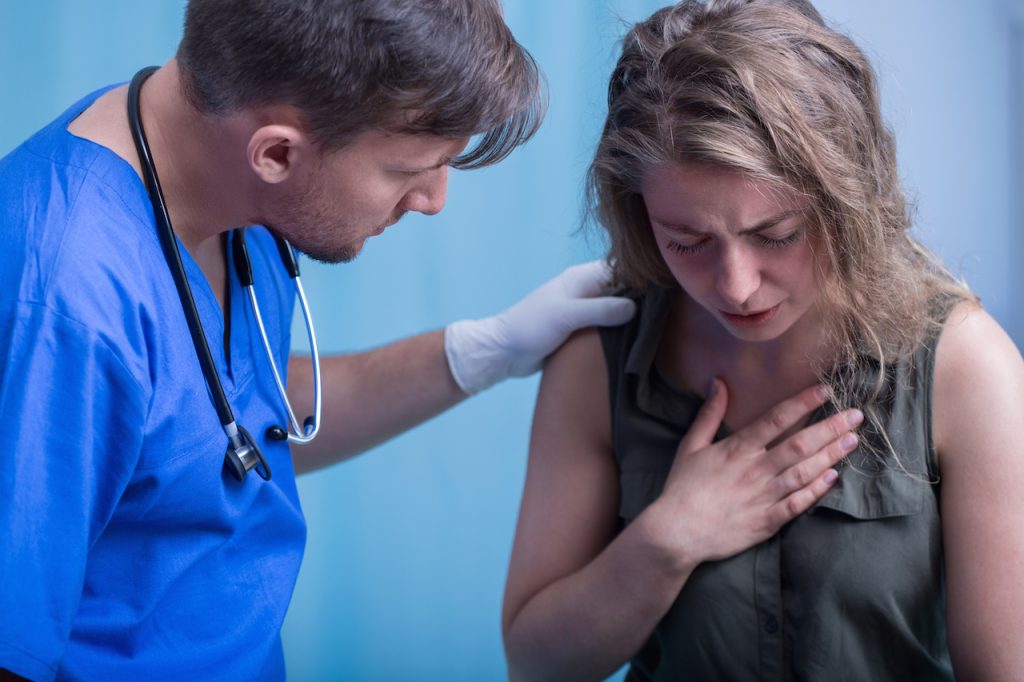
On Dec. 22, 2020, a patient at Decatur Morgan Hospital in Decatur, Alabama suffered a severe allergic reaction called anaphylaxis just minutes after receiving Pfizer/BioNTech’s experimental messenger RNA (mRNA) BNT162b2 vaccine for COVID-19.1 2 3 In a press release, the Alabama Department of Public Health (ADPH) stated the patient, who reportedly had a history of […]
Over 3,000 “Health Impact Events” After COVID-19 mRNA Vaccinations
Between Dec. 11 and 18, 2020, the U.S. Food and Drug Administration (FDA) granted Pfizer/BioNTech and Moderna pharmaceutical companies an Emergency Use Authorization (EUA)1 to distribute COVID-19 vaccines using messenger RNA (mRNA) technology that to date has not been licensed for use in humans.2 3 4 5 Although the Advisory Committee on Immunization Practices (ACIP) […]
Peruvian Man Suffers Guillain-Barré-like Symptoms After Receiving Sinopharm COVID-19 Vaccine

On Dec. 11, 2020, Peru’s National Institute of Health (INS) said it was temporarily suspending the Phase 1 clinical trial of an experimental COVID-19 vaccine developed by Sinopharm Group Co. Ltd. of Shanghai, China after one of the participants in the trial who got the vaccine showed difficulty in moving his arms and legs—symptoms of […]
CDC Shortens COVID-19 Quarantine Period to Seven Days

The U.S. Centers for Disease Control and Prevention (CDC) has revised its quarantine guidelines for people who may have been exposed to the SARS-CoV-2 virus.1 Since the COVID-19 pandemic declaration last winter, the agency has recommended a 14-day quarantine period that was based on estimates of the upper range of the incubation period for SARS-CoV-2 […]
Severe Allergic Reactions Reported After Pfizer/BioNTech COVID-19 Vaccinations

Last week the United Kingdom’s Medicines and Healthcare Products Regulatory Agency (MHRA) issued a strong warning to those who have a history of significant allergic reactions to medicines, foods or vaccines or who carry an epi-pen that they may be at increased individual risk for suffering a serious allergic reaction after receiving the Pfizer/BioNTech COVID-19 […]
COVID-19 Vaccine Trial Volunteer in India Develops Encephalopathy and Sues

A 40-year old business consultant in India, who volunteered to be a participant in clinical trials of the experimental COVID-19 vaccine (Covishield) created by AstraZeneca and Oxford University is suing Serum Institute of India (SII), which is producing the vaccine in India, for 50 million rupees ($680,000) after he developed acute encephalopathy (brain dysfunction) within […]

