Nordic Nations Suspend Use of Moderna COVID Shot Over Heart Inflammation Concerns

Following reports of an increased risk of myocarditis (inflammation of the heart muscle) and pericarditis (inflammation of the lining of the heart) in young adults after receiving Moderna/NIAID’s experimental messenger RNA (mRNA) COVID-19 biologic known as “Spikevax,” the governments of Denmark, Norway and Sweden have suspended the shot or will not make it available for […]
FDA’s Expert Panel Recommends COVID-19 Booster Dose for Seniors and Others, Rejects Booster Dose for Children

Despite not convening the Vaccines and Related Biologic Products Advisory Committee (VRBPAC) last month to vote on effectiveness and safety of the Pfizer-BioNTech COVID-19 vaccine (licensed under the name COMIRNATY), the U.S. Food and Drug Administration (FDA) convened the advisory committee on Friday, Sept. 17, 2021 to vote on booster doses of the vaccine.1 The […]
Robert Malone, MD on the Availability of Comirnaty

[Comirnaty] is absolutely not available. So the little trick that they have done here, is [the U.S. Food and Drug Administration] issued two separate letters for two separate vaccines. The Pfizer vaccine, which is what currently available is still under Emergency Use Authorization… The product that’s licensed [by the FDA] is the BioNTech product, which […]
Guillain-Barré Syndrome Warning Added to AstraZeneca’s Vaxzevria COVID Vaccine in Europe

The European Medicines Agency (EMA) last week added an autoimmune condition known as Guillain-Barré syndrome (GBS) as a possible side effect to the product information on AstraZeneca/Oxford University’s Vaxzevria (or “AZD1222”) experimental adenovirus vectored vaccine. GBS is a neurological disorder that causes pain, numbness, muscle weakness and sometimes paralysis.1 2 3 4 A total of […]
FDA Approves BioNTech’s Comirnaty. Pfizer COVID Shot Remains Experimental.
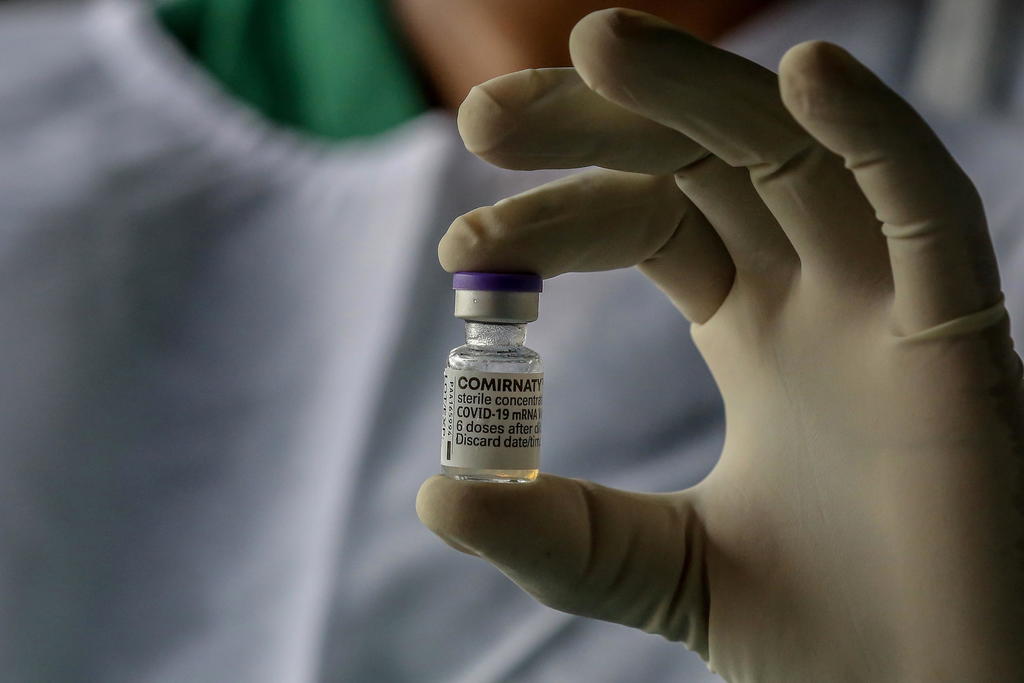
On Aug. 23, 2021, the U.S. Food and Drug Administration (FDA) gave full approval to a Biologics License Application (BLA) submitted by BioNTech Manufacturing GmbH of Mainz, Germany on May 18, 2021 for a biologic drug called COVID Vaccine, mRNA. The FDA gave permission to BioNTech to label the product “Comirnaty” and market it in […]
Canada Adds Bell’s Palsy Warning to Pfizer COVID Shot

Health Canada, the Canadian government’s department responsible for national health policy, earlier this month updated the product information label for Pfizer/BioNTech’s experimental messenger RNA (mRNA) biologic (also known as BNT162b2 or “Comirnaty” vaccine) to reflect reported cases of Bell’s palsy—an immune mediated neurological disorder characterized by temporary muscle weakness or paralysis on one side of […]

