3D Printed Vaccine Skin Patches Being Developed

In an attempt to increase consumer demand for COVID-19 shots, researchers have designed a vaccine printer that can print COVID messenger RNA (mRNA) skin patches in bulk, producing as many as 100 patches in 48 hours. The microneedle patches, which contain mRNA molecules in lipid nanoparticles, could pave the way for unlimited distribution of the […]
Smallpox Vaccine Manufacturers See Big Stock Price Increases After Monkeypox Outbreaks Reported

On May 26, 2022, U.S. Centers for Disease Control (CDC) officials held a press briefing and announced there had been nine cases of monkeypox identified in seven U.S. states and that two licensed smallpox vaccines were in the national stockpile and ready to be distributed.1 The CDC’s director said, “The U.S. has the resources we […]
Monkeypox Cases in U.S. Coincide With Vaccine Purchases by BARDA
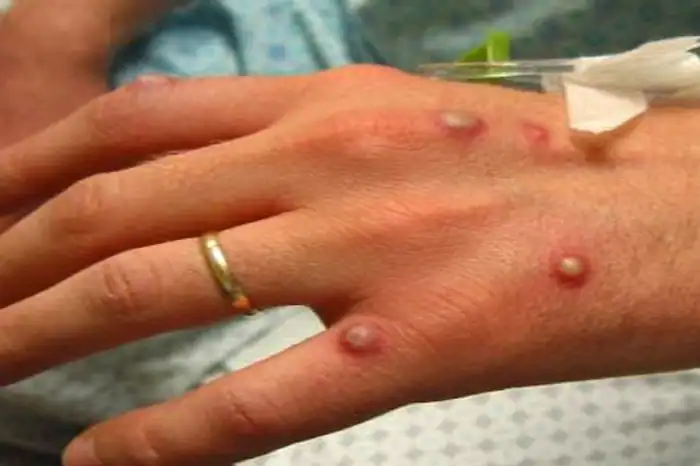
The World Health Organization (WHO) announced on May 21, 2022 that more than 100 confirmed or suspected cases of monkeypox had been identified in 12 countries, including between one and five confirmed cases in the U.S.1 Most of the cases have occurred in Europe with higher numbers in Portugal, Spain and the U.K. WHO officials […]
U.S. Taxpayers Paid Billions of Dollars for COVID-19 Vaccine Development

According to the U.S. Centers for Disease Control and Prevention (CDC), the federal government of the United States is providing COVID-19 vaccines free of charge to everyone five years and older living in the United States, regardless of their immigration or health insurance status.1 Although the CDC states that COVID vaccines are provided to the […]
20 Volunteers Suffer Serious Reactions to Johnson & Johnson’s COVID-19 Vaccine

On Sept. 23, 2020, U.S. pharmaceutical and medical devices company Johnson & Johnson, Inc. announced the start of the Phase 3 human clinical trial for its experimental Ad26.COV2.S vaccine for COVID-19. The New Jersey-based company, which is partnering with its subsidiary Janssen Pharmaceutica NV of Belgium on development of the vaccine, is planning to enroll […]
AstraZeneca Halts, Resumes COVID-19 Vaccine Trial After Serious Neurological Event
News update, Sept. 15, 2020—U.S. regulators paused AstraZeneca’s vaccine trial in the U.S. while the National Institutes of Health (NIH) launches an investigation of its own into a serious side effect in an unnamed patient in Britain. “The highest levels of NIH are very concerned,” said Dr. Avindra Nath, intramural clinical director and a leader […]

